|
Alloy Powder Cores feature slow flux saturation as field strength increased due to distributed gaps. An alloy is a mixture of metals that has bulk metallic properties different from those of its constituent elements. Alloys can be formed by substituting one metal atom for another of similar size in the lattice (substitutional alloys), by inserting smaller atoms into holes in the metal lattice (interstitial alloys), or by a combination of both. Although the elemental composition of most alloys can vary over wide ranges, certain metals combine in only fixed proportions to form intermetallic compounds. An intermetallic compound is a type of metallic alloy that forms an ordered solid-state compound between two or more metallic elements. G. E. R. Schulze in 1967 defined intermetallic compounds as solid phases containing two or more metallic elements, with optionally one or more non-metallic elements, whose crystal structure differs from that of the other constituents. Under this definition, the following are included:
Strukturbericht designation
In crystallography, a Strukturbericht designation is a system of detailed crystal structure classification by analogy to another known structure.
Each Strukturbericht designation is described by a single space group, but the designation includes additional information about the positions of
the individual atoms, rather than just the symmetry of the crystal structure. While Strukturbericht symbols (SBS) exist for many of the earliest
observed and most common crystal structures, the system is not comprehensive, and is no longer being updated. However, for most of our metal
alloy systems the crystal lattice structure D03 type of SBS is still well described by the designation.
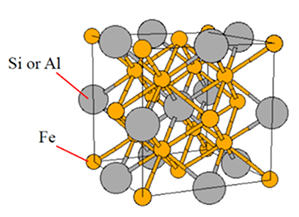
Figure 1. D03 structure: a body-centered cubic (BCC) derivative structure
Ferromagnetism
Ferromagnetism, a property of certain materials (such as iron) which results in a large observed magnetic permeability and high magnetization, is
the strongest type and responsible for the common phenomenon of magnetism in soft magnetics encountered in the electrical industry. Materials
which have magnetic properties similar to iron (e.g., nickel and cobalt and many alloys of these three elements) are ferromagnetic. In a landmark
paper in 1948, Louis Néel showed that there are two levels of magnetic alignment that result in this behavior. One is ferromagnetism in the strict
sense, where all the magnetic moments are aligned. The other is ferrimagnetism, where some magnetic moments point in the opposite direction
but have a smaller contribution, so there is still a spontaneous magnetization.
Amorphous (non-crystalline) ferromagnetic metallic alloys can be made by very rapid quenching (cooling) of a liquid alloy. These have the advantage that their properties are nearly isotropic (not aligned along a crystal axis); this results in low coercivity, low hysteresis loss, high permeability, and high electrical resistivity. Allotropes of iron and concerned alloy
At atmospheric pressure, three allotropic forms of iron exist, depending on temperature: alpha iron (α-Fe), gamma iron (γ-Fe), and delta iron (δ-Fe).
At 912 °C and below, the crystal structure is the BCC, called α-iron allotrope. α-iron features low magnetocrystalline anisotropy (κ) and
magnetostriction constant (λs) while its low resistivity limits the applications towards high frequency range.
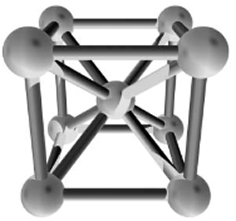
Figure2. BCC structure of α-iron
From a practical standpoint, a high resistivity is usually desirable to decrease the eddy-current losses in the material and thereby decrease the
power wasted. It is disclosed by R. M. Bozorth (Ferromagnetism, 2003) that dissolving a small amount of one element in another increases the
resistivity (ρ) of the latter. The following figure 3 (source: Ferromagnetism, 2003 by Bozorth) shows the relative effects of various elements, the
common binary alloys of iron. Obviously, the addition of silicon and aluminum to iron leads to essential increment on resistivity. It then comes out
various type of alloy systems, such as Fe-Si, Fe-Si-Al, Fe-Ni, Fe-Ni-Mo and others to be applied to magnetic powder core industry. The usefulness
of iron-silicon alloys was made known to the world as the result of researches begun by Hadfield in 1882. This led to many investigations by
others, and especially to the report on magnetic properties by Barrett, Brown, and Hadfield in 1900. And it was the 1900 paper which sets the
stage for commercial use.
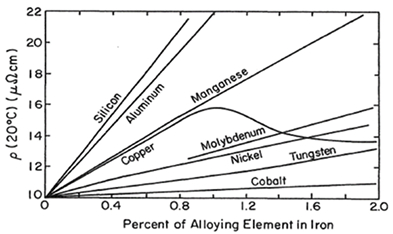
Figure 3. Dependence of resistivity on the addition of small amounts of various elements to iron
Some remarkable properties of alloys in Fe-Si-Al ternary system have been uncovered by Masumoto of Tohoku university at Sendai Japan, who
investigated specimens containing up to 14% aluminum and 14% silicon. The diagram of figure 4 (source: Ferromagnetism, 2003 by Bozorth) shows
lowest magnetocrystalline anisotropy (κ) and magnetostriction constant (λs), which are found to be extraordinarily high of permeability near the
composition 9.5-10% silicon, 5-5.5% aluminum. The material of high permeability is quite brittle, is easily reduced to powder (dust), and is
appropriately named Sendust.
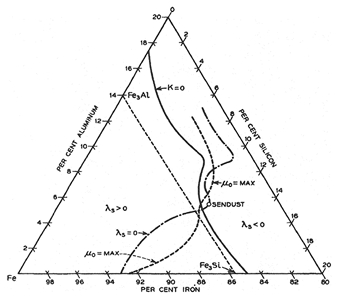
Figure 3. Dependence of resistivity on the addition of small amounts of various elements to iron
Due to the fact that around 15% of silicon and aluminum dissolved in iron it gives rise to the resistivity of sendust being increased largely so as to
reduce eddy current loss in a large scale. As a result of that, sendust cores bring the application frequency of alloy powder cores into the range of
higher than 50KHz, the major stream of switching power supply units.
The first comprehensive study of the iron-nickel alloys in weak and medium fields was begun in 1913 by Elmen who also discovered a special heat treatment ("Permalloy treatment") that increased still further the initial permeabilities of the alloys from 50-90% nickel. Another investigation was begun independently by Yensen in 1915. By heat-treating the Fe-50%Ni alloy in hydrogen Yensen in 1925 attained about the same maximum permeability as Elmen found in 78 Permalloy (Fe-78%Ni) and distinct improvements in hysteresis loss. The use of hydrogen annealing raises the maximum permeability of the 45 or 50% alloy by a factor of 2 to 5, depending on the temperature and on the care taken to avoid contamination of the material during the fabrication and heat treatment. The more recent improvements in the properties of the magnetically soft iron-nickel alloys have been brought about by the control of impurities and using certain heat treatments. The following figure 5 (source: Ferromagnetism, 2003 by Bozorth) shows the variation of saturation magnetization with temperature for several binary Fe-Ni alloy systems. 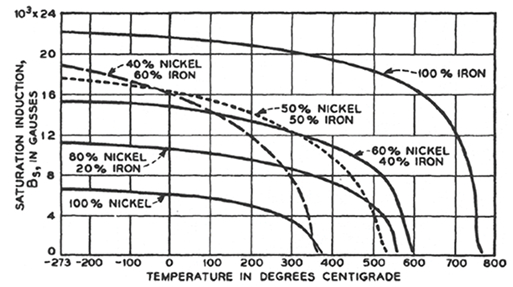
Figure 5. Dependence of saturation induction of various iron-nickel alloys on the temperature
R&D in ACME
The R&D activity in ACME electronics on alloy powder cores can be dated in early 2015. During that period the target product types were mini-drum
cores made in Fe-Si-Cr alloy powders for SMD power inductors. To further extent the product service to the customers, the research done in
the company on the other types of alloy powders and amorphous/nanocrystalline powder preparation was involved. As the success of hybrid/
composite core development in combination with the excellent low loss properties of our power ferrites, the focus on Fe-Si-Al, Fe-Si and Fe-Ni
powder cores has then become the best strategy to confront the future magnetic design challenges.
|